Healthcare Building Codes and Design After COVID-19
By Mark Stefanak and Karpinski Engineering | Jun 16, 2020
Key Takeaways
- Screening areas may become a standard part of the healthcare building environment.
- Hospitals need operating rooms that are ready to accommodate highly infectious patients. This can be accomplished by adding a negatively-pressured anteroom (whether temporary or permanent). We anticipate an FGI requirement calling for hospitals to have airborne infectious isolation operating rooms.
- Hospitals need to be able to keep highly infectious patients separate from non-infectious or immune-compromised patients. A code change allowing for a “pandemic mode” would give hospitals more control over air pressurization and ventilation when facing a surge of highly-infectious patients.
- Relative humidity levels impact the spread of viruses. The committee overseeing ASHRAE Standard 170: Ventilation of Health Care Facilities may take a fresh look at humidification requirements and their impact on patient wellbeing.
As healthcare engineers who supported hospitals and healthcare organizations through the first wave of the COVID-19 pandemic, we’ve seen a lot in the past couple months that has influenced our thought process.
As we look to the future, we’re thinking about how hospital buildings can support infection control even more than they do now. Here are four ways we think that healthcare codes and standards may change as a result of the pandemic.
Screening Areas Become the Norm
Currently, we’ve seen hospitals screening anyone coming into the building, taking people’s temperatures and verifying what they’ve been exposed to. In states like Ohio, part of re-opening any business includes a requirement for employee self-screening.
We wouldn’t be surprised if screening areas become a standard part of the healthcare building environment, like security booths. You walk in, sign in, answer some questions, and have a no-contact temperature scan. This would apply to employees, patients and visitors.
Hospitals Create Operating Rooms to Accommodate Highly Infectious Patients
COVID-19 brought an atypical scenario to the forefront: the need to operate on patients with a highly infectious disease, possibly an airborne infection.
While operating rooms are positively pressurized in relationship to the spaces around them, airborne infectious isolation (AII) rooms are negatively pressurized. Now we’re seeing healthcare organizations asking about making adjustments to their ORs to operate on a highly infectious patient.
From a design standpoint, this could be accomplished via an anteroom that is at a negative pressure with respect to the OR and adjacent space. The anteroom could be permanent or temporary (using modular panels).
- For a temporary anteroom, the idea is that all the core systems would already be in place: the bulkhead in the ceiling for the wall panels, a dedicated supply terminal unit for the anteroom, a closeable return terminal unit for the OR, and an exhaust fan to serve both the OR and anteroom. When needed, the hospital can install the modular panels to create the anteroom and change the operating mode of the HVAC system.
- As part of cleaning and sterilizing an airborne infectious isolation OR, the hospital would want to leave the room empty to allow the air to change. They would also want to review their cleaning and sterilization procedures for returning the OR to typical use.
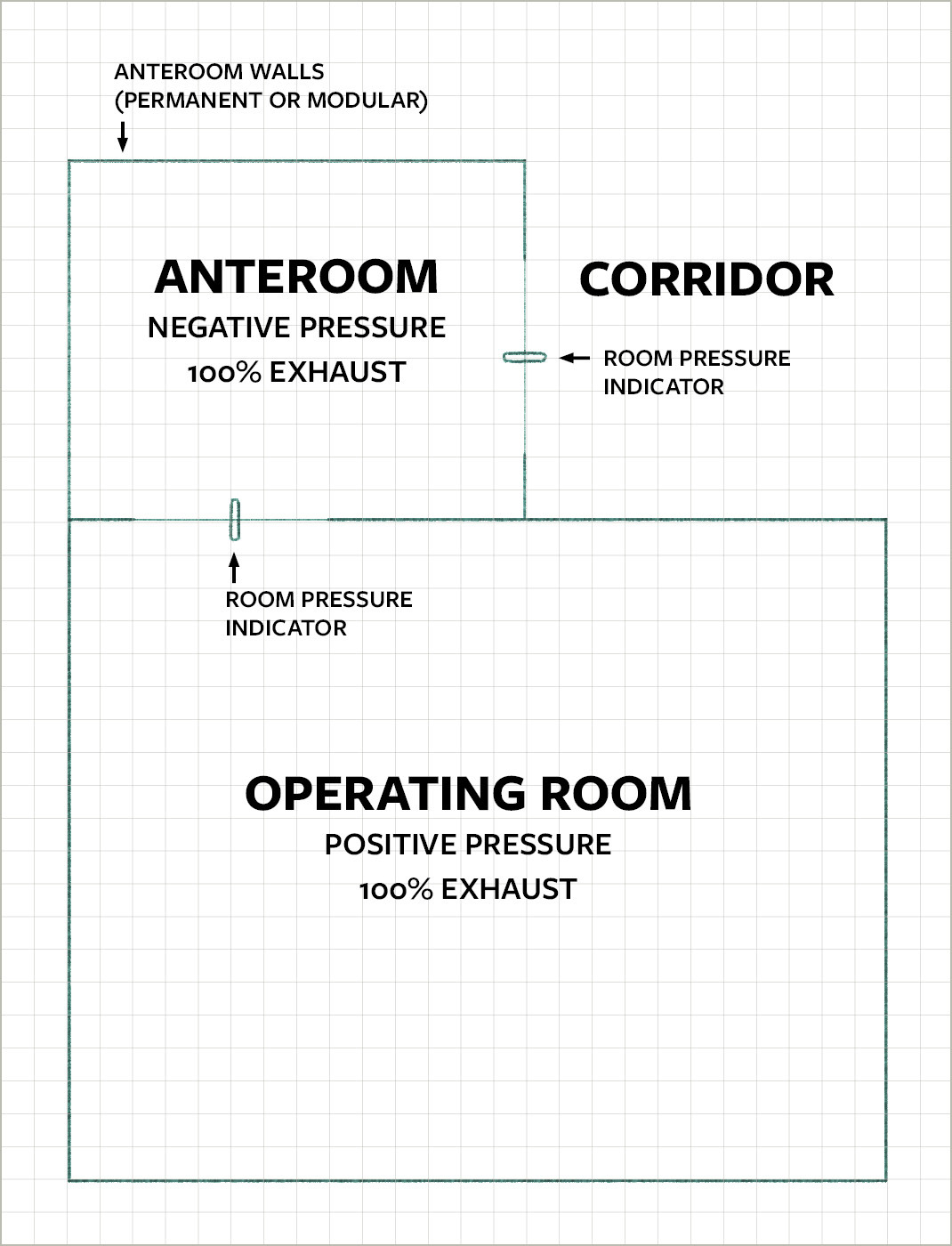
We anticipate an FGI requirement for hospitals to have airborne infectious isolation operating rooms: ORs that are 100% outside air and have anterooms to allow for these types of procedures.
From a logistics standpoint, we envision two ways this requirement could take shape:
- One operating room per facility is to have an anteroom (whether permanent or temporary) and the capability to switch to 100% exhaust.
- As part of an Infection Control Risk Assessment (ICRA) Plan, one hospital in a region becomes the designated location for these ORs, allowing separation of highly-infectious patients from immune-compromised patients. The hospital might have multiple airborne infectious isolation ORs available (again, either permanent or temporary).
Hospitals Gain More Flexibility and Control of Air Pressurization and Ventilation
Hospitals don’t generally have a whole wing of AII rooms, but as we’ve seen, it could become necessary to accommodate a surge of highly infectious patients and keep them separate from non-infectious or immune-compromised patients.
For example, a hospital might want to take an entire patient wing that’s typically returning air, and instead make it 100% outside air and at negative pressure in relationship to the rest of the building.
Perhaps a code change would require a “pandemic mode.” It would allow equipment to be sized for the worst-case scenario (running at 100% outside air) and hospitals could switch to this mode of operation during a flu, COVID, or similar situation.
Hospital Humidification Requirements Will Get a Fresh Look
Research shows that too low of relative humidity increases the transmission of viruses. In its COVID-19 healthcare resources, ASHRAE suggests healthcare facilities consider maintaining relative humidity at 40-60%. (For additional information on building humidification, see the reference list at the end of this ASHRAE white paper.)
ASHRAE Standard 170: Ventilation of Health Care Facilities contains humidification requirements for spaces such as operating rooms, treatment rooms, and intensive care. However, the Standard does not provide minimum relative humidity requirements for standard patient rooms and numerous other rooms in a hospital.
We might see those responsible for developing ASHRAE 170 start to ask questions like: Does relative humidity have an impact on patient wellbeing? If so, what do we want to do about it? Should ASHRAE 170 contain additional minimum relative humidity requirements?
The COVID-19 pandemic has confronted our healthcare system with new challenges and priorities. As healthcare designers, our responsibility is to develop new solutions to support patients and caregivers on the road to healing.